First-Ever 3D View Shows How Killer T Cells Destroy Cancer
Scientists have captured the first three-dimensional view of the moment killer T cells strike cancer cells, revealing a meticulously organized contact zone that allows them to destroy dangerous cells without harming healthy neighbors.
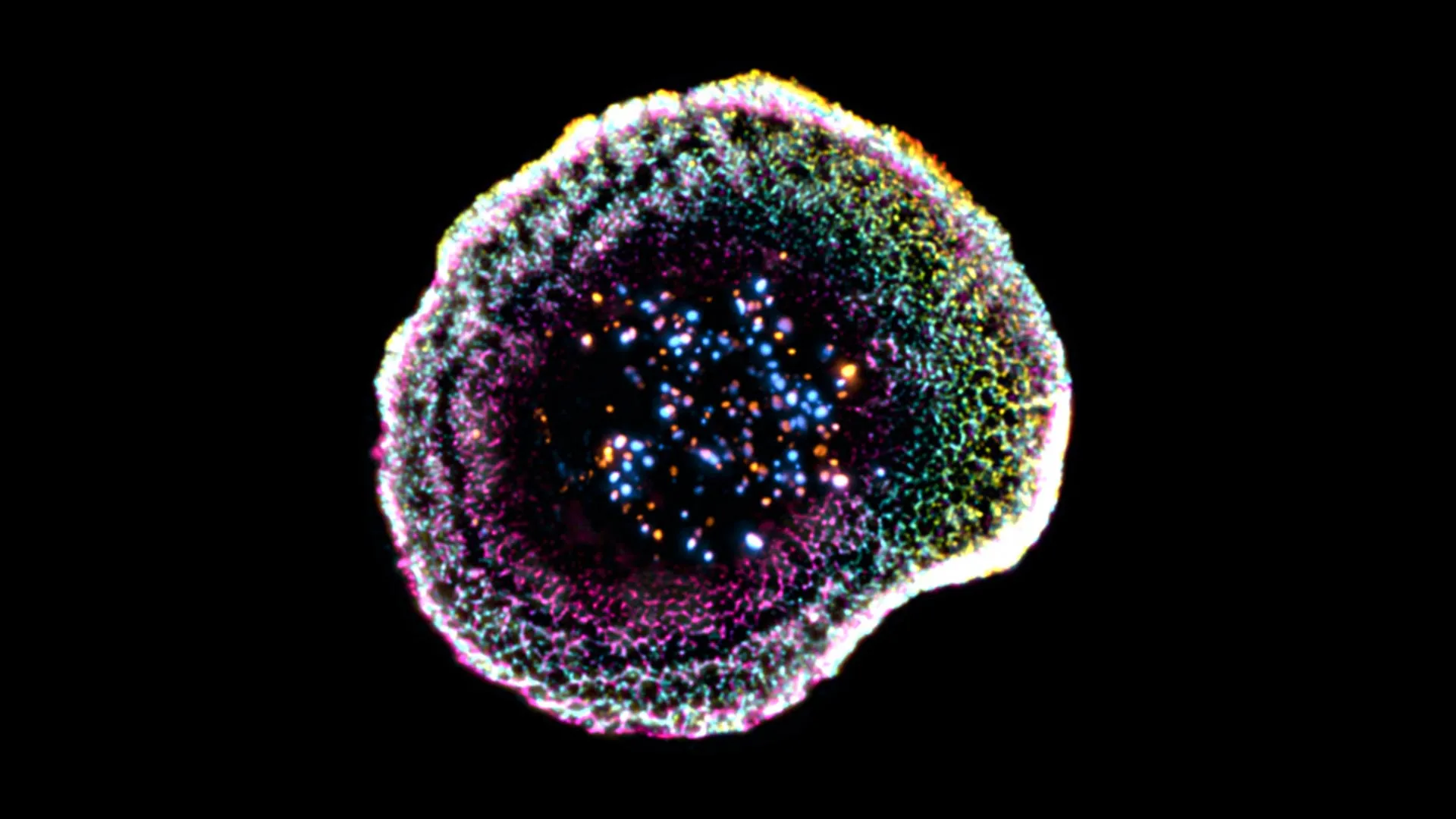
The breakthrough, published today in Nature, shows that T cells form a tiny, highly structured interface—like a molecular bullseye—that delivers lethal proteins directly into cancer cells with astonishing precision.
What This Means for Cancer Treatment
“This is the first time we’ve seen the actual geometry of the killing machinery,” said Dr. Elena Marchetti, lead researcher at Stanford University’s Institute for Immunology. “It’s not a random attack—it’s a choreographed strike.”
The 3D imagery reveals that T cells lock onto targets using an array of surface proteins, then deploy a ring-shaped complex called the “immunological synapse” to punch holes in the cancer cell membrane. This prevents collateral damage to surrounding tissue.
Background
Killer T cells, also known as cytotoxic T lymphocytes, are a key component of the adaptive immune system. They patrol the body, identifying and destroying cells infected by viruses or transformed into tumors.
Until now, scientists could only observe this process in two dimensions, which limited understanding of the spatial organization. The new technique uses cryo-electron tomography and advanced computational modeling to reconstruct the interface in 3D at near-atomic resolution.
How the Research Was Done
The team engineered model cancer cells and exposed them to human T cells. Using rapid freezing to preserve the moment of engagement, they captured thousands of images and assembled them into a 3D movie.
“We could literally see the T cell’s killing machinery aligning like magnets,” said co-author Dr. James Huang, a biophysicist at the University of California, San Francisco. “The precision is breathtaking.”
Implications for Immunotherapy
Immunotherapies that boost T cell activity—such as checkpoint inhibitors and CAR-T cell therapy—have revolutionized cancer treatment. However, many patients still don’t respond, and side effects like autoimmune reactions remain a problem.
“Understanding this molecular choreography could help us design smarter therapies,” Marchetti said. “If we can improve the accuracy of the strike, we might make treatments more effective and less toxic.”
What This Means
This discovery provides a target for drug developers: enhancing the formation of the immunological synapse could turn weak T cells into efficient killers. Conversely, preventing overactivity might reduce side effects.
“We now have a roadmap of the killing zone,” Huang added. “It’s like having a blueprint for a guided missile.”
Next Steps
The team plans to investigate how cancer cells sometimes evade the T cell attack—for instance, by disrupting the synapse formation. That could lead to new ways to reverse resistance.
“We’re already working on CRISPR screens to identify genes that control the synapse structure,” Marchetti said. “That could open up entirely new avenues for cancer therapy.”
Original Source
Read the original study at Nature.